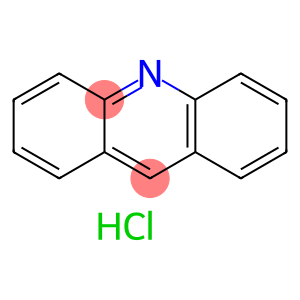
Acridine hydrochloride
Acridine hydrochloride
CAS: 17784-47-3
Molecular Formula: C13H10ClN
Acridine hydrochloride - Names and Identifiers
| Name | Acridine hydrochloride |
| Synonyms | ACRIDINIUM CHLORIDE Acridine hydrochlori Acridine hydrochloride ACRIDINE HYDROCHLORIDE acridin-10-ium chloride acridine,monohydrochloride Acridine HydrochlorideHydrate |
| CAS | 17784-47-3 |
| EINECS | 241-762-6 |
| InChI | InChI=1/C13H9N.ClH/c1-3-7-12-10(5-1)9-11-6-2-4-8-13(11)14-12;/h1-9H;1H |
Acridine hydrochloride - Physico-chemical Properties
| Molecular Formula | C13H10ClN |
| Molar Mass | 215.68 |
| Melting Point | 250-255°C (dec.) |
| Boling Point | 346.7°C at 760 mmHg |
| Flash Point | 153.8°C |
| Vapor Presure | 0.000113mmHg at 25°C |
| Appearance | powder to crystal |
| Color | Light yellow to Brown |
| Merck | 14,122 |
| Storage Condition | Inert atmosphere,Room Temperature |
| MDL | MFCD00035149 |
Acridine hydrochloride - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R22 - Harmful if swallowed |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S22 - Do not breathe dust. |
| WGK Germany | 3 |
| RTECS | AR9322000 |
| HS Code | 29339980 |
Acridine hydrochloride - Reference Information
| use | pyridine hydrochloride as an organic reagent can be used to test the content of cobalt, iron, zinc and other elements. |
| preparation | nitrobenzene is prepared from benzene by nitration, aniline is prepared by reduction, acridine is prepared by acylation, shrinkage and closed loop, and the target compound acridine is synthesized by salt formation with hydrogen chloride gas. |
| category | toxic substances |
| toxicity classification | poisoning |
| acute toxicity | subcutaneous-mouse LD50: 300 mg/kg |
| flammability hazard characteristics | flammability; heating decomposition releases toxic nitrogen oxides and hydrogen chloride smoke |
| storage and transportation characteristics | warehouse ventilation and low temperature drying |
| fire extinguishing agent | dry powder, foam, sand, carbon dioxide, mist water |
Last Update:2024-04-09 19:05:10
Supplier List
Spot supply
Product Name: Acridine hydrochloride Visit Supplier Webpage Request for quotationCAS: 17784-47-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Acridine hydrochloride Request for quotation
CAS: 17784-47-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 17784-47-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Acridine hydrochloride Visit Supplier Webpage Request for quotation
CAS: 17784-47-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 17784-47-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Acridine hydrochloride Visit Supplier Webpage Request for quotationCAS: 17784-47-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Acridine hydrochloride Request for quotation
CAS: 17784-47-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 17784-47-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Acridine hydrochloride Visit Supplier Webpage Request for quotation
CAS: 17784-47-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 17784-47-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


